Your goal is to empower your team with the information they need to be productive, safe and informed on the job. Our online safety course library contains more than 1,500 training topics to help improve employee performance and provide self-paced training that is engaging, consistent and measurable. UL Solutions’ team of health and safety experts and advisers carefully develop the education plans to target the diverse needs of learners. You’ll find more than 1,000 employee health and safety course titles and more than 400 life sciences regulatory course titles in our proprietary course library.
Online Employee Health and Safety (EHS) Training
Your goal is to empower your team with the information they need to be productive, safe, and informed on the job.
Our online safety course library contains more than 1,500 training topics to help improve employee performance and provide self-paced training that is engaging, consistent and measurable.
Embrace Intelligent Learning with ComplianceWire® LMS
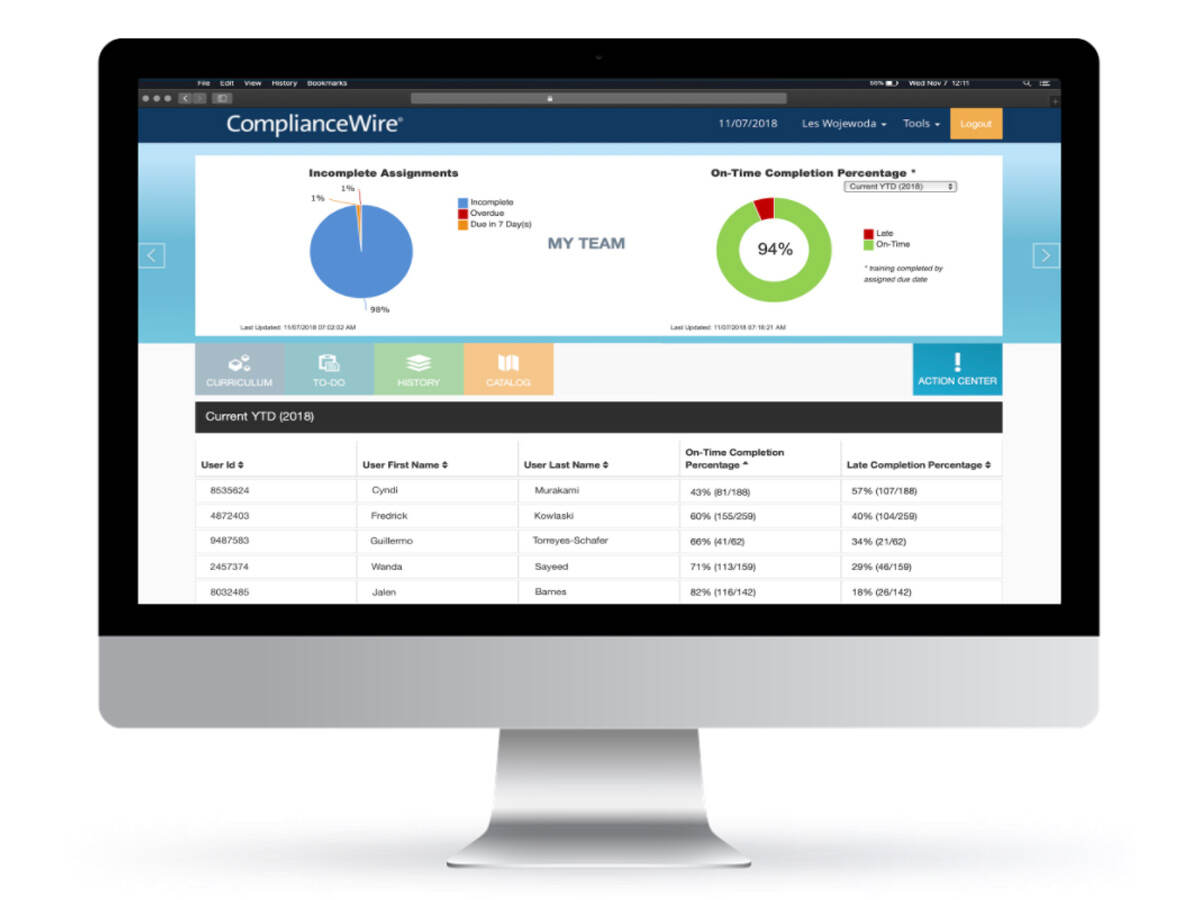
ComplianceWire®, the best-in-class solution for regulated industries, is a powerful, Part 11 compliant and fully validated Learning Management System (LMS) used by the US FDA and other global regulators and relied on by Pharmaceutical, Medical Device and Biologics companies to help improve productivity and reduce risk.
EHS Training and Learning Management
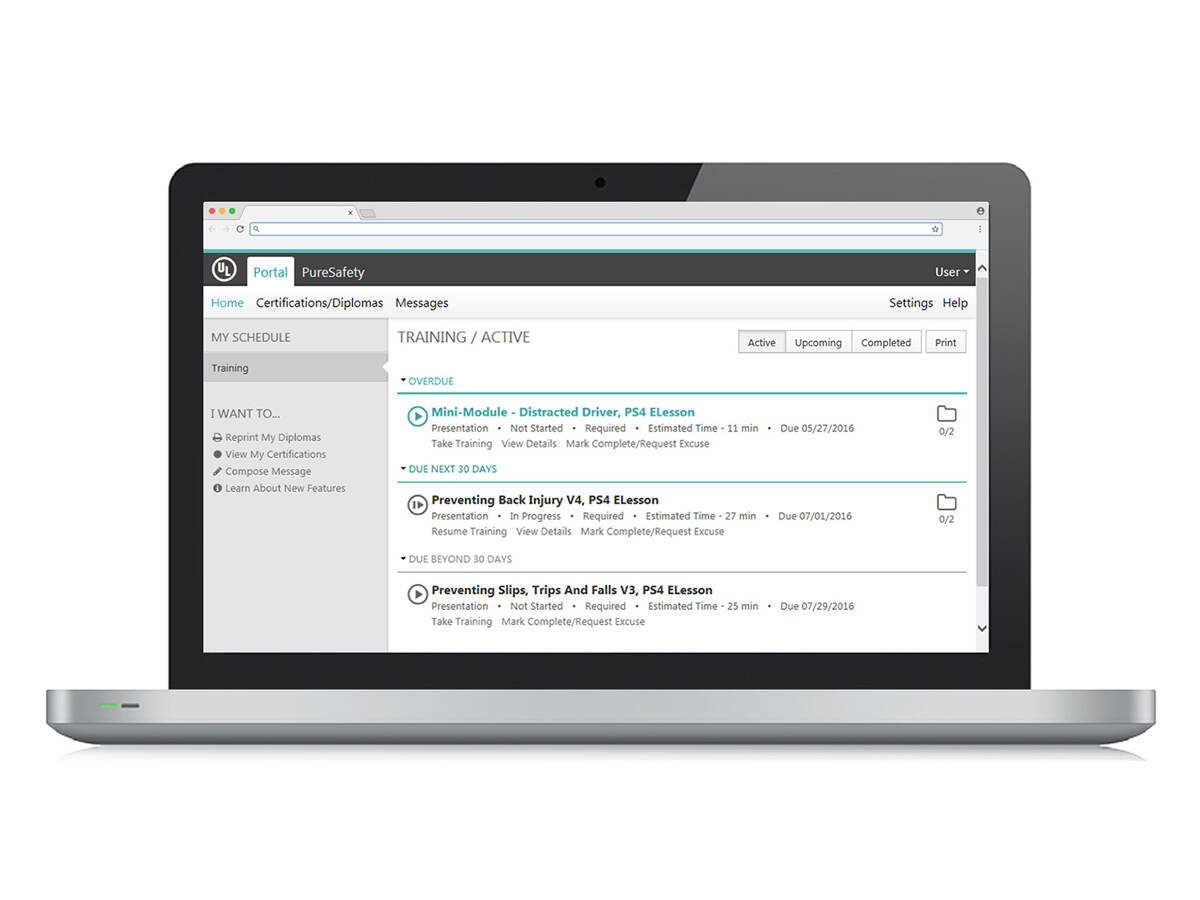
Effective workplace training demands more than content with a pretty dashboard. Get access to training courses that are updated on a regular basis, keeping them fresh and aligned with evolving regulations. The extensive library covers safety, life sciences, healthcare, and general industry topics including incident management, bloodborne pathogens, hazard communication, and OSHA 10 and 30 hour courses (for U.S.). You can trust your employees are getting the knowledge and instruction they need to stay safe in the workplace.
Enterprise Learning Management
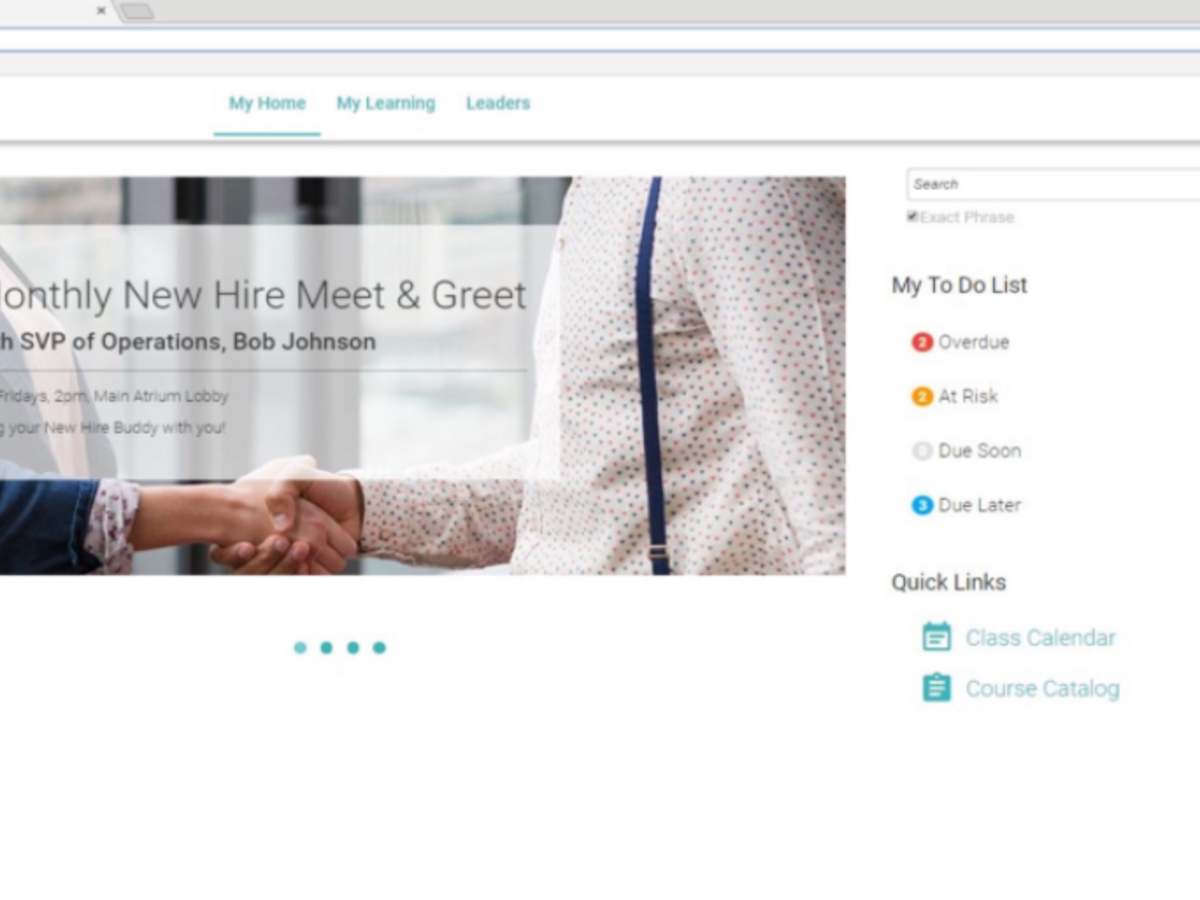
Learnshare Enterprise Learning Management System: Our enterprise system enables flexible learning and development capabilities that support today’s best practices for compliance, development and performance management across the diverse needs of growing organizations.
eLearning Content
Professionally developed education programs, using industry experts, that carefully target the diverse needs of learners. Our proprietary library of 1000+ courses includes over 1500 EHS course titles and over 400 Life Sciences regulatory course titles (many of which are co-developed with the US FDA).
Custom content for unique learning programs
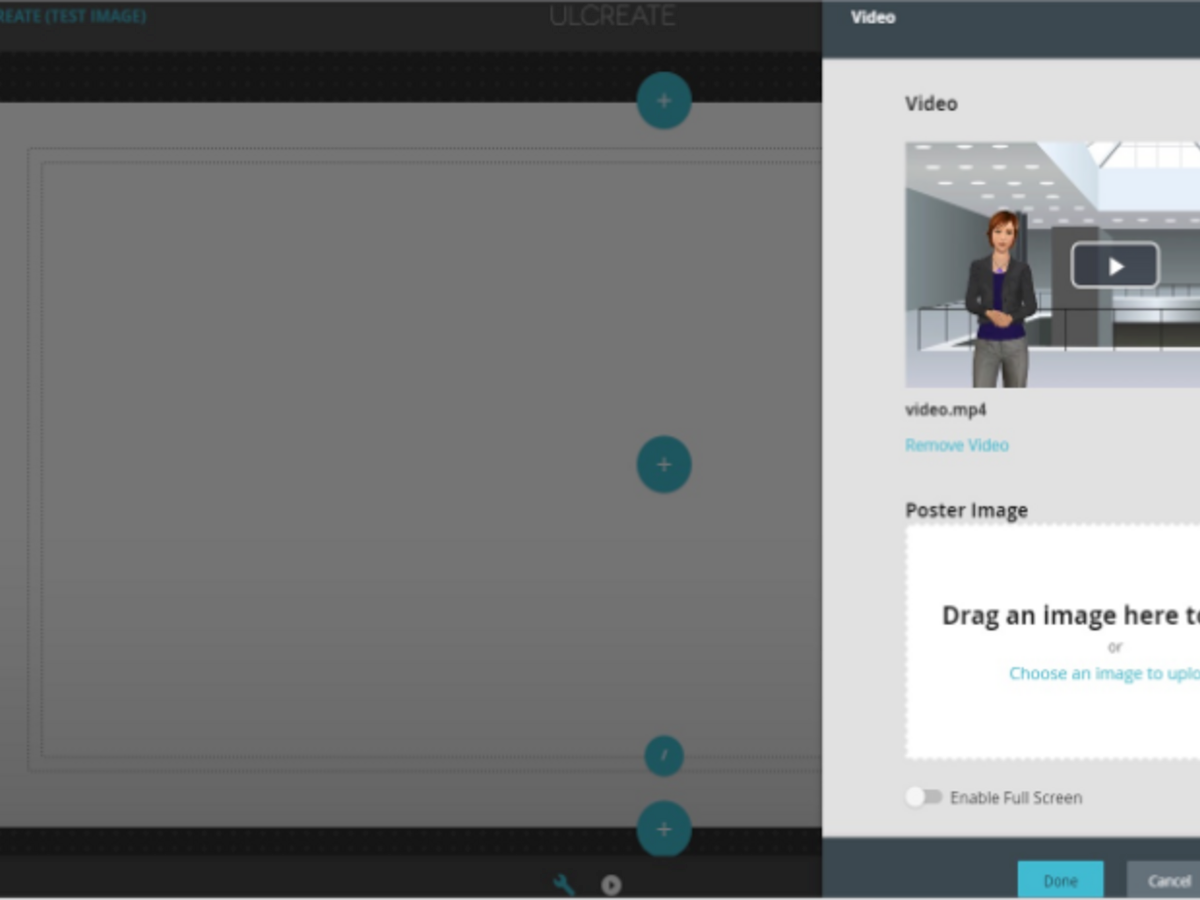
With UL Solutions CREATE self-authoring tool and a team of adult learning and design specialists, we can help bring your learning needs to life. Our team of experts can adapt existing learning content or create brand new learning experiences to fit your unique needs.
UL Solutions Named a Top Employee Health, Safety and Wellness Training Company by Training Industry
Training Industry recognizes UL Solutions’ comprehensive online training content that helps empower employees with the skills and knowledge they need to succeed and stay safe on the job.